Give yourself the freedom to more easily manage your diabetes with Tandem Diabetes Care mobile apps.
Freedom to Deliver
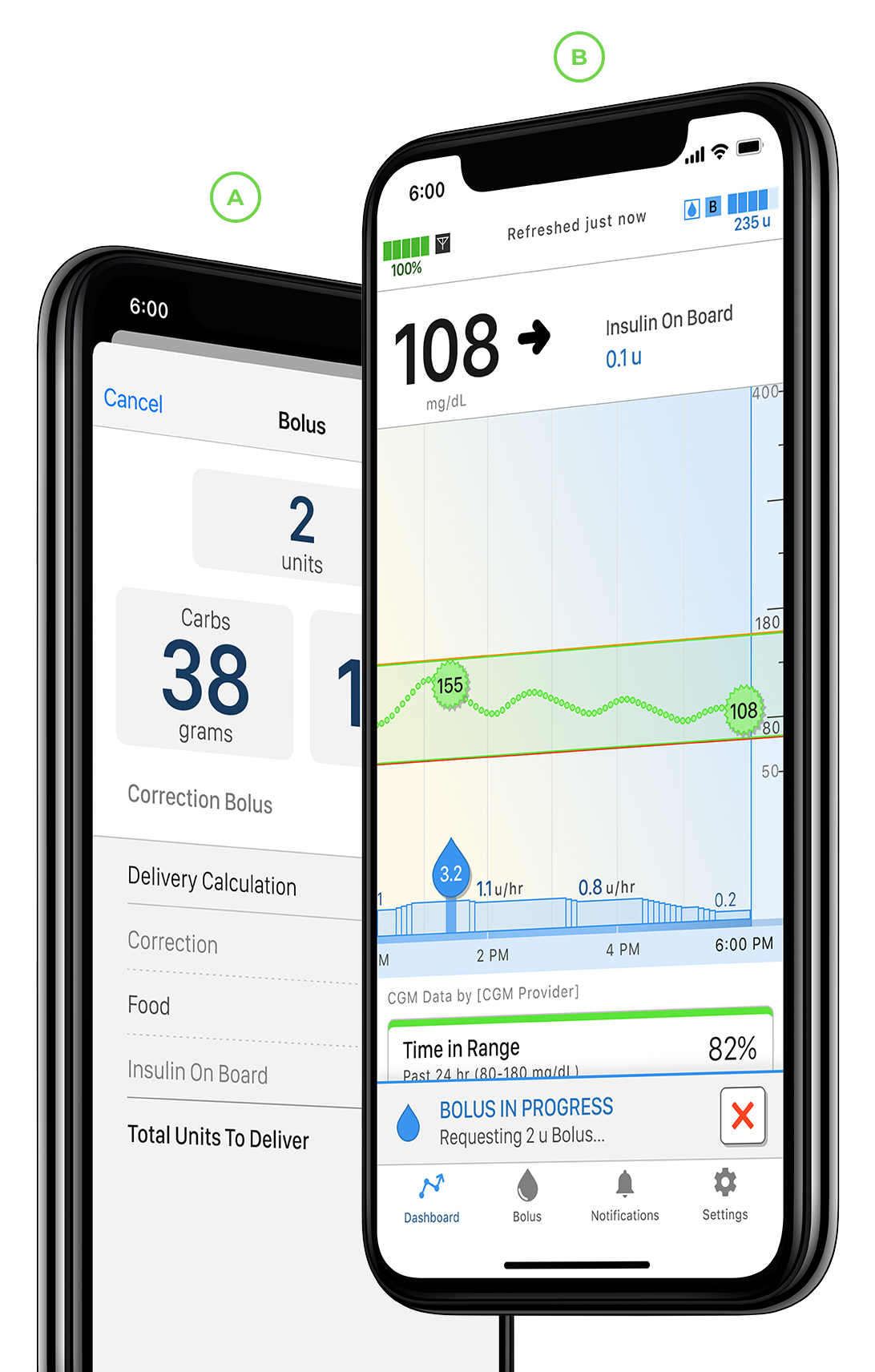
When you pair a Tandem mobile app to your Tandem insulin pump, you have the freedom to bolus from your compatible smartphone* without touching your pump.
Freedom to See Clearly
Conveniently view your pump data, including basal and bolus events, insulin on board, carbs, current settings, and both pump and sensor status directly on your smartphone.
Freedom to be in the Know
Wirelessly upload your pump and therapy data to the cloud-based Tandem Source platform and allow your authorized healthcare provider(s) easy online access.†
Full iPhone Control with the Tandem Mobi System
Tandem Mobi is the small, automated insulin delivery system that’s controlled with a compatible, personal iPhone,‡ offering next-level comfort, convenience, and discretion. This is innovative diabetes management.
Meet MobiMobile App Features
The features available within the Tandem mobile apps are dependent on the version of your Tandem Mobi or t:slim X2 insulin pump, along with the model and operating system of your smartphone. See which app features are compatible with your device.
| App Features | Tandem Mobi Mobile App for Tandem Mobi Pump | Tandem t:slim Mobile App for t:slim X2 Pump |
|---|---|---|
| Bolus From Your Smartphone | ||
| View Pump Data | ||
| Receive Alerts & Alarms§ | ||
| Wirelessly Upload Pump Data | ||
| Stop & Resume Insulin Delivery | Only from | |
| Enable Temp Basal Rates | ||
| Manage Personal Profiles | ||
| Manage CGM Sensor Integration | ||
| Program Sleep Schedules | ||
| Enable Exercise Activity | ||
| Extended Bolus Delivery |
What's next?
Whether you're ready to get a Tandem insulin pump, or looking to check insurance coverage, you’ll find everything you need here.
